 
For the previous version, please go here. (iv) What is the nature of binding force b/w atoms and molecules which accounts the existence of matter in three states, i.e. In the year 1808, the English scientist and chemist John Dalton proposed Dalton’s atomic hypothesis, a scientific theory on the nature of matter. This is an updated version of our Atomic Theory I module. (iii) Why do atoms of the same or different elements combine at all to form molecules (ii) It could not explain why atoms of different elements have different masses, sizes, valencies, etc. By 1807, however, references to this work appeared in Daltons notebooks, and Daltons contemporaries viewed his atomic theory as a way of explaining why compounds combine in definite proportions. 
(i) It could explain the laws of chemical combination by mass but failed to explain the law of gaseous volumes. John Dalton was not familiar with Richters work when he developed his atomic theory in 1803. The atoms of an element are alike in all. Atoms can neither be created nor be destroyed. The main drawbacks of Dalton's Atomic Theory are: Matter consists of very small and indivisible particles called atoms. However, the subatomic particles do not represent the element. According to the current knowledge, subatomic particles like electrons, protons, and neutrons compose the atoms. But the brilliant researches conducted in the beginning of 20th century by Sir J.J Thomson, Lord Rutherford, Neils Bohr and others have revolutionised our knowledge about the structure of atom. Dalton's atomic theory is the basis of the current atomic theory, though the atoms are no more considered ‘indivisible.’. Atoms of different elements vary in size, mass, and chemical behavior. All atoms of the same element are identical and have the same mass. The five main points are: Matter is made up of atoms, small and indivisible particles. 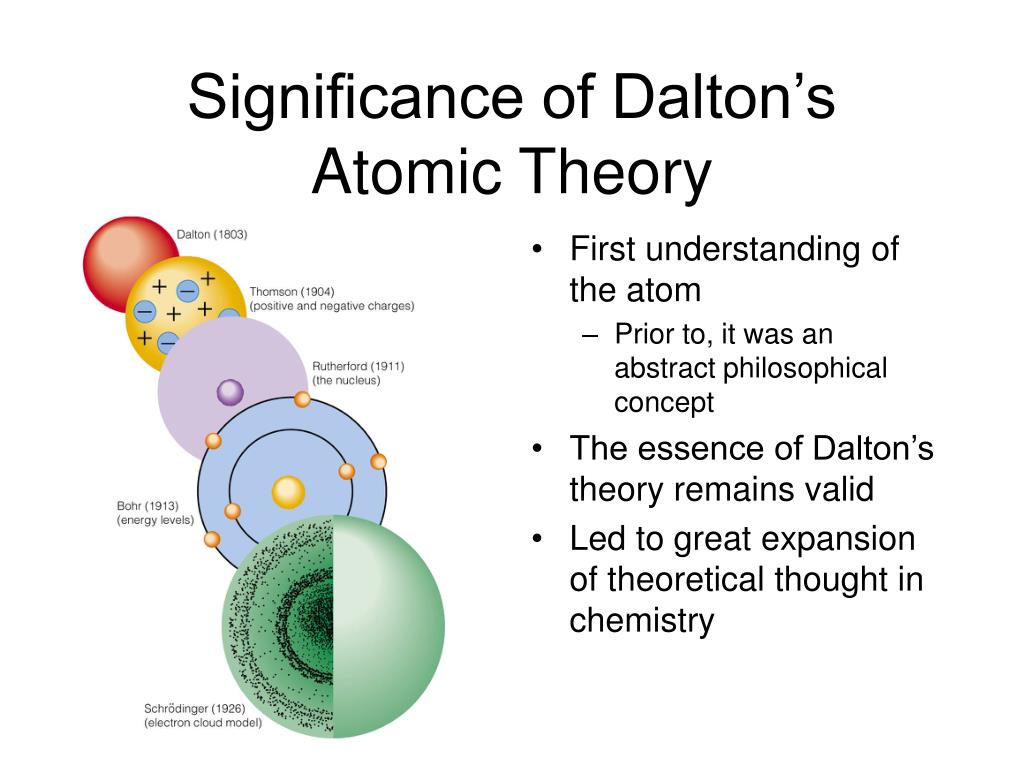
His atomic theory may be summarized as below. Daltons atomic theory proposed that an atom was the smallest indivisible and indestructible building block of matter. Dalton's Atomic Theory was formulated by John Dalton in 1808, and it remains a fundamental tenet of chemistry to this day. It gave a powerful initiative to the scientists about the study of matter during 19th century. (b) Dalton’s at omic theory At the beginning of the 19th century, atomic theory as a philosophy of matter was already well developed by Dalton who developed his atomic theory based on the role of atoms during chemical reactions. Dalton's atomic theory was the first milestone towards the inner structure of matter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
